Breast Implant Illness: Understanding the Symptoms, Evidence, and Treatment Considerations
The majority of patients with silicone breast implants, whether for cosmetic or reconstructive purposes, do not report any complications (American Society of Plastic Surgeons, 2023). However, a small percentage of individuals with breast implants have reported experiencing a range of symptoms, including brain fog, fatigue, anxiety, depression, hair loss, brittle nails, and joint pain. This collection of symptoms is commonly referred to as Breast Implant Illness (BII). It is important to note that BII is not linked to any specific type of implant or manufacturer (U.S. Food and Drug Administration, 2022).
Historical Context and Scientific Investigations
Concerns regarding systemic health issues related to breast implants are not new. In the early 1990s, similar symptoms were reported, prompting a thorough investigation in 1998 by the Independent Review Group (IRG) in the UK. The IRG concluded that there was no evidence linking silicone gel implants to any long-term systemic illness (IRG Report, 1998). More recently, the Institute of Medicine (IOM, 1999) and various global health authorities, including the FDA, have also reviewed data and found no definitive connection between silicone breast implants and chronic illness.
Challenges in Diagnosing Breast Implant Illness
One of the significant challenges in diagnosing Breast Implant Illness (BII) is that its symptoms overlap with a variety of other medical conditions, such as autoimmune disorders, chronic fatigue syndrome, and hormonal imbalances (American Society for Aesthetic Plastic Surgery, 2023). Since no standardized diagnostic criteria exist for BII, patients often face uncertainty regarding their symptoms and treatment options.
Despite growing awareness, particularly through social media platforms, no conclusive scientific evidence has established a direct causal relationship between silicone breast implants and systemic illnesses (FDA, 2023). One study even reported that women without breast implants exhibited symptoms similar to those attributed to BII, further complicating the discussion (Watad et al., 2017).
The Role of Implant Removal and Capsulectomy
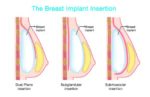
Some patients choose to undergo implant removal (explant surgery) with or without total capsulectomy in an attempt to alleviate their symptoms. However, the effectiveness of this approach remains unproven. To date, there is no scientific evidence supporting that en-bloc capsulectomy, total capsulectomy, or partial capsulectomy significantly improves BII symptoms (De Boer et al., 2021).
Additionally, capsulectomy carries risks, including increased surgical complications, longer scars, and potential breast contour irregularities. The decision to undergo surgery should be carefully weighed against these risks, with realistic expectations regarding symptom resolution.
The Importance of Evidence-Based Care
While anecdotal reports and patient advocacy groups have increased awareness of Breast Implant Illness, plastic surgeons and medical professionals must base their approach on scientific evidence rather than social media-driven narratives. Patient safety remains the top priority. Plastic surgeons should:
- Listen with compassion to patients reporting BII symptoms.
- Educate patients on the current scientific understanding of BII.
- Discuss all available options—including monitoring, medical evaluation for other underlying conditions, and surgical removal when appropriate.
- Emphasize informed decision-making, ensuring patients understand both the benefits and risks associated with explant surgery.
Conclusion
Breast Implant Illness remains a topic of ongoing research, and while current evidence does not support a direct causal link between silicone breast implants and systemic illness, medical professionals must continue to investigate patient concerns. The role of healthcare providers is to guide patients through evidence-based care, helping them make informed decisions while prioritizing safety.
By maintaining a scientific approach, open communication, and patient-centered care, plastic surgeons can ensure that those with breast implants receive accurate information and the highest standard of treatment.
References
- American Society of Plastic Surgeons (ASPS). (2023). Breast Implant Safety and Research. https://www.plasticsurgery.org
- U.S. Food and Drug Administration (FDA). (2022). Breast Implant Safety. https://www.fda.gov
- Independent Review Group (IRG). (1998). Safety of Silicone Breast Implants. UK Government Report.
- Institute of Medicine (IOM). (1999). Safety of Silicone Breast Implants. National Academies Press.
- Watad, A. et al. (2017). “Autoimmune Syndrome Induced by Adjuvants (ASIA) – A New Clinical Entity.” Clinical Rheumatology.
- De Boer, M. et al. (2021). “Breast Implant Illness: A Narrative Review.” Aesthetic Surgery Journal.